By Jen Christensen
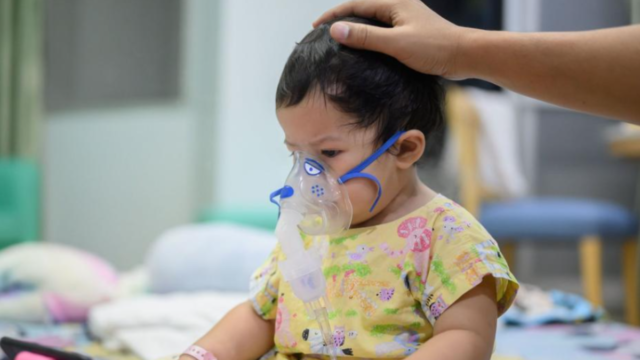
The US Centers for Disease Control and Prevention said Friday it is recommending a new vaccine to help protect babies against severe respiratory syncytial virus, or RSV.
“This is another new tool we can use this fall and winter to help protect lives,” said CDC Director Dr. Mandy Cohen. “I encourage parents to talk to their doctors about how to protect their little ones against serious RSV illness, using either a vaccine given during pregnancy, or an RSV immunization given to your baby after birth.”
The CDC’s independent vaccine advisers voted Friday to recommend the maternal vaccine.
The vote to recommend the vaccine was 11 to 1. The specific question advisers voted on was: “Maternal RSV vaccine is recommended for pregnant people during 32 through 36 weeks gestation, using seasonal administration, to prevent RSV lower respiratory tract infection in infants.”
The vaccine, named Abrysvo and made by Pfizer, would be given in a single dose to pregnant people late in their pregnancy. The RSV season is typically September through January in most parts of the US. Mothers of infants born outside of RSV season would not get the vaccine, but a new RSV prevention antibody, called nirsevimab, would be recommended to protect those infants.
The committee also voted 11 to 1 on a resolution that would add the RSV vaccine in the Vaccines for Children program. The CDC told the committee Friday that next week it was going to update its vaccine schedule to include all the independent committee’s recommendations this year about vaccines, including this one.
Typically the vaccine schedule is published in February and it fails to capture the most recent recommendations from this committee. The experts said it’s important to keep the schedule current, because it is often what insurance companies use to decide what vaccines it will cover. It’s also important for clinicians, since they rely on it to know what vaccines they need to give their patients. There are two vaccine schedules: one that makes recommendations for adults and another that makes suggestions for children and adolescents.
The RSV vaccine could make a real difference for this country’s health, experts on the committee said.
“Throughout my career, this has been a difficult disease with just supportive care treatment available, because there have been no options. So today is an exciting day,” said committee member Dr. Katherine Poehling, professor of pediatrics and epidemiology and prevention, Wake Forest School of Medicine, who voted for the vaccine.
Studies show that the vaccine would provide infants protection against RSV lower respiratory tract disease and severe lower respiratory tract disease for at least the first six months of their lives.
In August, the US Food and Drug Administration approved the vaccine, voting unanimously that the vaccine was effective and safe. In a trial with more than 7,000 pregnant people and their infants, the vaccine cut the risk that infants needed to see a doctor or be admitted to the hospital. Side effects were minor. The most common side effects, felt by less than 10% of participants were pain at the injection site, headache, muscle pain, and nausea, Pfizer said.
In the trials the vaccine was administered to pregnant people even earlier than what the CDC independent committee is recommending, but the experts wanted to give the vaccine in the 32 to 36 week of the pregnancy to avoid the risk of extreme preterm birth. A trial of a similar RSV vaccine from GSK was stopped because of a preterm birth risk.
Another adjustment was the price of the vaccine. Pfizer told the committee that the cost of the vaccine would be about $295 per dose, which was nearly 50% higher than the projected price they presented in June. Some CDC committee members questioned why the vaccine was so expensive. By comparison the Tdap vaccine that is given to pregnant people costs about $46 to $52. Studies show the cost of the new RSV vaccine would likely be one of the biggest barriers for people to get the vaccine.
“I’m really very disappointed about the expense of this vaccine, especially when a recommendation may be made to provide it to all pregnant women,” committee member Dr. Jamie Loehr, the owner of Cayuga Family Medicine in Ithaca, New York said.
Chair of the committee Dr. Grace Lee, a professor of pediatrics at Stanford University, called the vaccine “an excellent option to protect infants from hospitalization,” but she also added that it is “one of the more expensive interventions we have recommended.”
Vaccine coverage among pregnant people has dropped in general since the pandemic, according to the CDC, so it is not totally clear how many people will get this new vaccine. If the vaccine was universally accepted, Pfizer told the committee that this newly approved shot would prevent up to 16,000 hospitalizations and more than 300,000 RSV-related doctor visits each year.
This is not the only option to protect infants from RSV. For years, pre-term infants have received antibodies that can help protect them from infection, but that was just for children considered vulnerable to the most serious kinds of infections. This year the FDA and CDC also approved an antibody treatment that can be given to all infants after birth called nirsevimab.
The group did not feel that infants needed both nirsevimab and maternal vaccination. Either would be recommended. There are advantages to getting either, according to a presentation given to the committee by the CDC. A vaccine provides protection immediately after birth, it may be more resistant to virus mutation, and using a maternal vaccine avoids the need to inject the infant. There is, however, a small risk of preterm birth with the vaccine, but giving the vaccine to the pregnant person at 32 weeks or later reduces this risk.
The benefits of nirsevimab over the vaccine, studies show, is that protection might wane more slowly than the protection from the vaccine.
The treatment also provides antibodies directly to the infant, and there is no risk that the antibody treatment would cause a preterm birth since it is given directly to the infant, rather than to the pregnant person. The treatment is, however, more expensive than the vaccine. It costs $495, but the cost should be covered by insurance. Another disadvantage of the treatment is that it may also not be available, since supplies of nisevimab have been limited.
Dr. Brenda Hughes, a representative from the American College of Obstetricians and Gynecologists (ACOG) told the committee that it is “unequivocally supportive” of a full recommendation for the maternal RSV vaccine for pregnant people.
“This RSV vaccine is efficacious and could decrease the risk of severe disease in many infants. And while nirsevimab is clearly highly efficacious, it may not be available or may not be preferred by a parent or healthcare facility as the primary intervention,” Hughes told the committee.
The FDA has required Pfizer to conduct post-marketing studies to assess if the shot can lead to preterm birth or hypertensive disorders in pregnancy, including pre-eclampsia.
Pfizer said it plans to do a large trial after the vaccine is officially on the market. They’ll use a large database of commercial claim data to try and evaluate safety endpoints. The CDC also plans to use V-Safe, a program developed specifically to monitor Covid-19 vaccine safety that has been redesigned to track other people’s post-vaccine experience with other vaccines.
In the rare chance that someone had a bad reaction to the shot they can also report it to the Vaccine Adverse Event Reporting System.
“Today’s ACIP recommendation for maternal immunization with Abrysvo reinforces the wide-ranging impact vaccines can have, including helping protect infants immediately at birth from the potentially severe and life-threatening complications that can develop from RSV,” said Pfizer’s Dr. Luis Jodar, the company’s chief medical affairs officer for vaccines, antivirals and evidence generation.








You must be logged in to post a comment Login